4.2 Lipids
Learning Objectives
By the end of this section, you will be able to:
- Describe the four major types of lipids
- Explain the role of fats in storing energy
- Differentiate between saturated and unsaturated fatty acids
- Describe phospholipids and their role in cells
- Define the basic structure of a steroid and some steroid functions
- Explain how cholesterol helps maintain the plasma membrane’s fluid nature
Lipids include a diverse group of compounds that are largely nonpolar in nature. This is because they are hydrocarbons that include mostly nonpolar carbon–carbon or carbon–hydrogen bonds. Nonpolar molecules are hydrophobic (“water fearing”), or insoluble in water. Lipids perform many different functions in a cell. Cells store energy for long-term use in the form of fats. Mammals store fats in specialized cells, or adipocytes, where fat globules occupy most of the cell’s volume. Plants store fat or oil in seeds and use them as a source of energy during seedling development. Lipids also provide insulation from the environment for plants and animals (Figure 4.9). For example, they help keep aquatic birds and mammals dry when forming a protective layer over fur or feathers because of their water-repellant hydrophobic nature. Lipids are also the building blocks of many hormones and are an important constituent of all cellular membranes. Lipids include fats, oils, waxes, phospholipids, and steroids.

Fats and Oils
A fat molecule consists of two main components—glycerol and fatty acids. Glycerol is an organic compound (alcohol) with three carbons, five hydrogens, and three hydroxyl (-OH) groups. Fatty acids have a long chain of hydrocarbons to which a carboxyl group is attached, hence the name “fatty acid.” The number of carbons in the fatty acid may range from 4 to 36. The most common are those containing 12–18 carbons. Some fatty acids have common names that specify their origin. For example, palmitic acid, a saturated fatty acid, is derived from the palm tree. Arachidic acid is derived from Arachis hypogea, the scientific name for groundnuts or peanuts. In a fat molecule, the fatty acids attach to each of the glycerol molecule’s three carbons with an ester bond through an oxygen atom to form a triacylglycerol, also known as triglyceride (Figure 4.10). During this ester bond formation, three water molecules are released. The three fatty acids in the triacylglycerol may be similar or dissimilar.
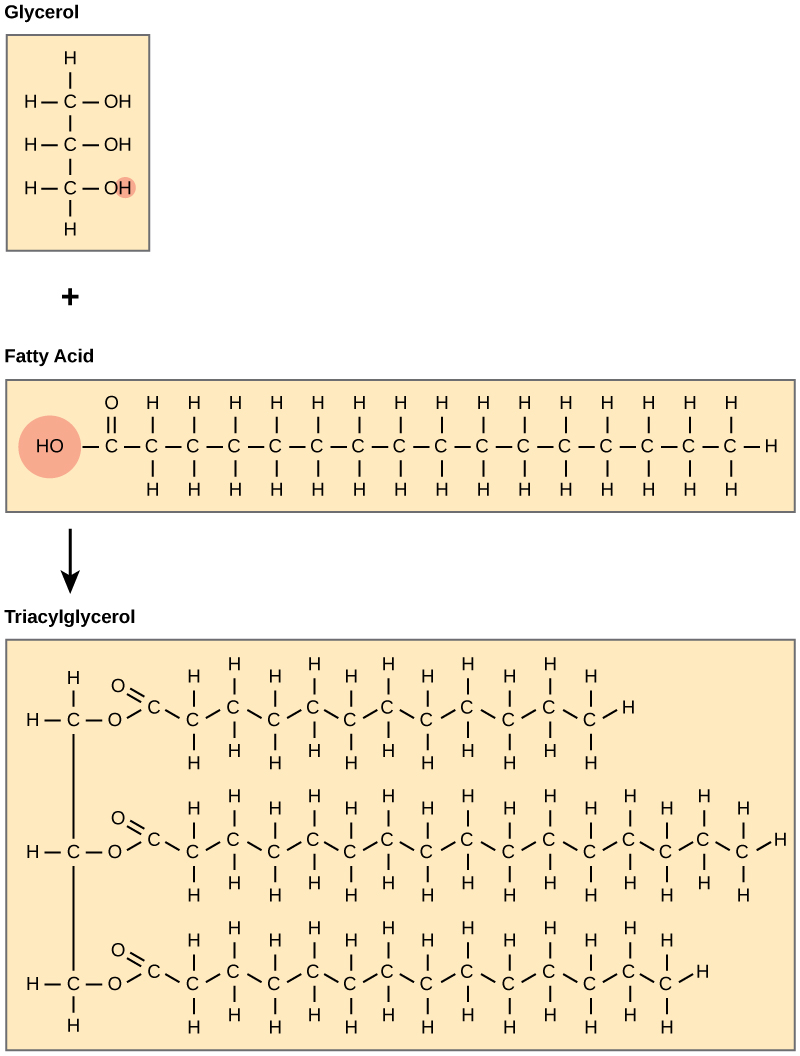
Saturated Fatty Acids
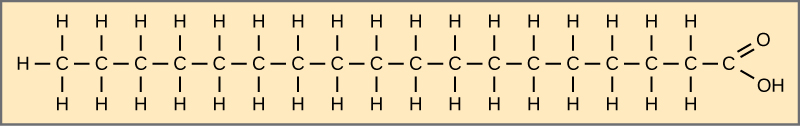
Fatty acids may be saturated or unsaturated. In a fatty acid chain, if there are only single bonds between neighboring carbons in the hydrocarbon chain, the fatty acid is saturated. Saturated fatty acids are saturated with hydrogen. In other words, the number of hydrogen atoms attached to the carbon skeleton is maximized. Stearic acid is an example of a saturated fatty acid (Figure 4.11). Animal fats with stearic acid and palmitic acid (common in meat) and the fat with butyric acid (common in butter) are examples of saturated fats. Saturated fats are solid at room temperature and usually of animal origin.
Unsaturated Fatty Acids
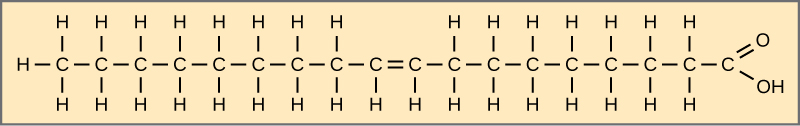
When the hydrocarbon chain contains a double bond, the fatty acid is unsaturated. Oleic acid is an example of an unsaturated fatty acid (Figure 4.12). If there is one double bond in the molecule, then it is a monounsaturated fatty acid, and if there is more than one double bond, then it is a polyunsaturated fatty acid.
The carbon-carbon double bonds in unsaturated fatty acids can be in either the cis- or trans-configuration, as Figure 4.13 illustrates. If hydrogen atoms are present in the same side of the carbon-carbon double bond, it is a cis-fatty acid. If the hydrogen atoms are on different sides, it is a trans-fatty acid. The cis-double bond, causes a bend in the carbon backbone of the fatty acid means that the triglyceride molecules cannot pack tightly, so they remain liquid at room temperature. We use the word “oil” to denote fats that are liquid at room temperature. In contrast, saturated fats have a linear carbon backbone, which allows for tight packing of fatty acid chains and is why saturated fats are solid at room temperature. Olive oil, corn oil, canola oil, and cod liver oil are examples of unsaturated fats. Unsaturated fats help to lower blood cholesterol levels; whereas, saturated fats contribute to plaque formation in the arteries.
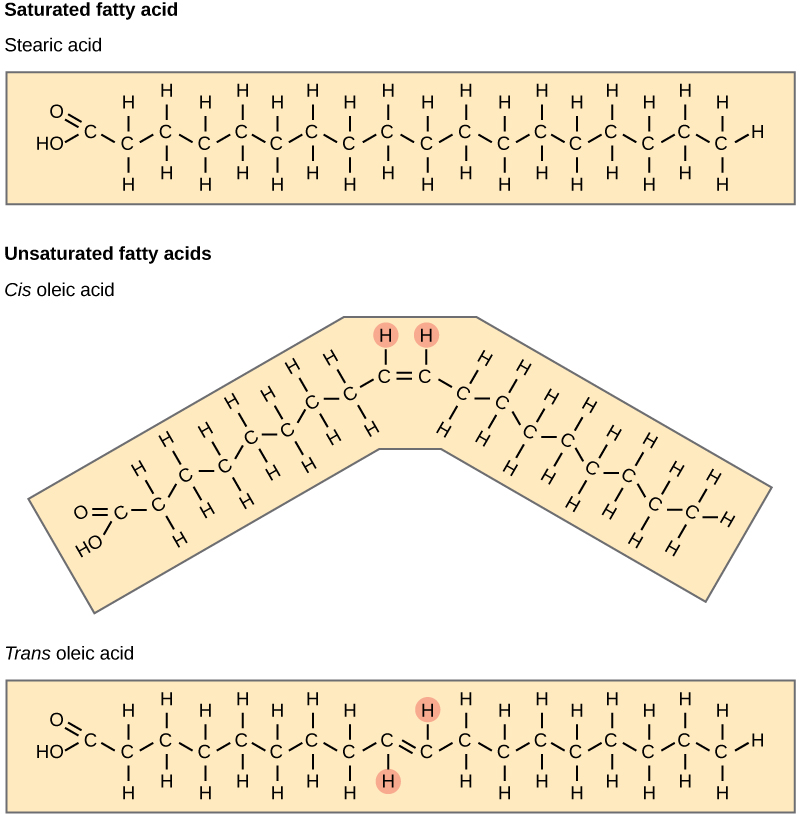
Alternatively, triglycerides with trans-double bonds (popularly called trans fats), have relatively linear fatty acid chains that are able to pack tightly together at room temperature and form solid fats. The food industry chemically hydrogenates oils to make them semi-solid and of a consistency desirable for many processed food products. Simply speaking, hydrogen gas is bubbled through oils to solidify them. Margarine, some types of peanut butter, and shortening are examples of artificially hydrogenated trans fats. During this hydrogenation process, double bonds of the cis-configuration in the hydrocarbon chain may convert to double bonds in the trans-configuration.
Recent studies have shown that an increase in trans fats in the human diet may lead to higher levels of low-density lipoproteins (LDL), or “bad” cholesterol, which in turn may lead to plaque deposition in the arteries, resulting in heart disease. Many food manufacturers have recently banned using trans fats, and food labels are required to display the trans fat content.
Omega Fatty Acids
Polyunsaturated fatty acids have multiple double bonds in the hydrocarbon chain. The farthest carbon away from the carboxyl group is numbered as the omega (ω) carbon, and if the double bond is between the third and fourth carbon from that end, it is an omega-3 fatty acid. Essential fatty acids are those that the human body requires but does not synthesize. Omega-3 fatty acids (like those in Figure 4.14) fall into this category and are one of only two known for humans (the other is omega-6 fatty acid). Consequently, they have to be supplemented through ingestion via the diet.
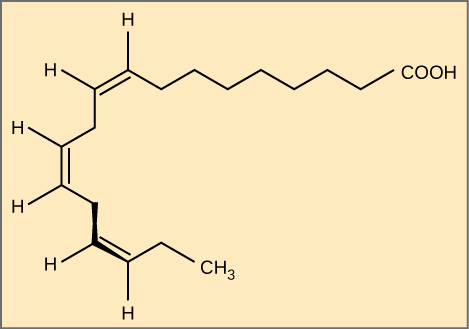
Nutritionally important because the body does not make them, omega-3 fatty acids include alpha-linoleic acid (ALA), eicosapentaenoic acid (EPA), and docosahexaenoic acid (DHA), all of which are polyunsaturated. Salmon, trout, and tuna are good sources of omega-3 fatty acids. Research indicates that omega-3 fatty acids reduce the risk of sudden death from heart attacks, lower triglycerides in the blood, decrease blood pressure, and prevent thrombosis by inhibiting blood clotting. They also reduce inflammation, and may help lower the risk of some cancers in animals.
Like carbohydrates, fats have received considerable bad publicity. It is true that eating an excess of fried foods and other “fatty” foods leads to weight gain. However, fats do have important functions. Many vitamins are fat soluble, and fats serve as a long-term storage form of fatty acids: a source of energy. They also provide insulation for the body. Therefore, we should consume “healthy” fats in moderate amounts on a regular basis.
Waxes
Wax covers some aquatic birds’ feathers and some plants’ leaf surfaces. Because of waxes’ hydrophobic nature, they prevent water from sticking on the surface (Figure 4.15). There is no clear chemical definition of wax because waxes are variable mixtures of hydrocarbons, alcohols, aldehydes, ketones, and esters, among other molecules. The common feature is that the molecules that make up wax all have long hydrocarbon chains and are hydrophobic.

Phospholipids
Phospholipids are major constituents of the cells’ outermost layer, the plasma membrane. Like fats, they are comprised of fatty acid chains attached to a glycerol or sphingosine backbone. However, instead of three fatty acids attached as in triacylglycerol, there are two fatty acids forming diacylglycerol, and a modified phosphate group occupies the glycerol backbone’s third carbon (Figure 4.16). A phosphate group alone attached to a diacylglycerol does not qualify as a phospholipid. It is phosphatidate (diacylglycerol 3-phosphate), the precursor of phospholipids. An alcohol modifies the phosphate group. Phosphatidylcholine and phosphatidylserine are two important phospholipids that are in plasma membranes.
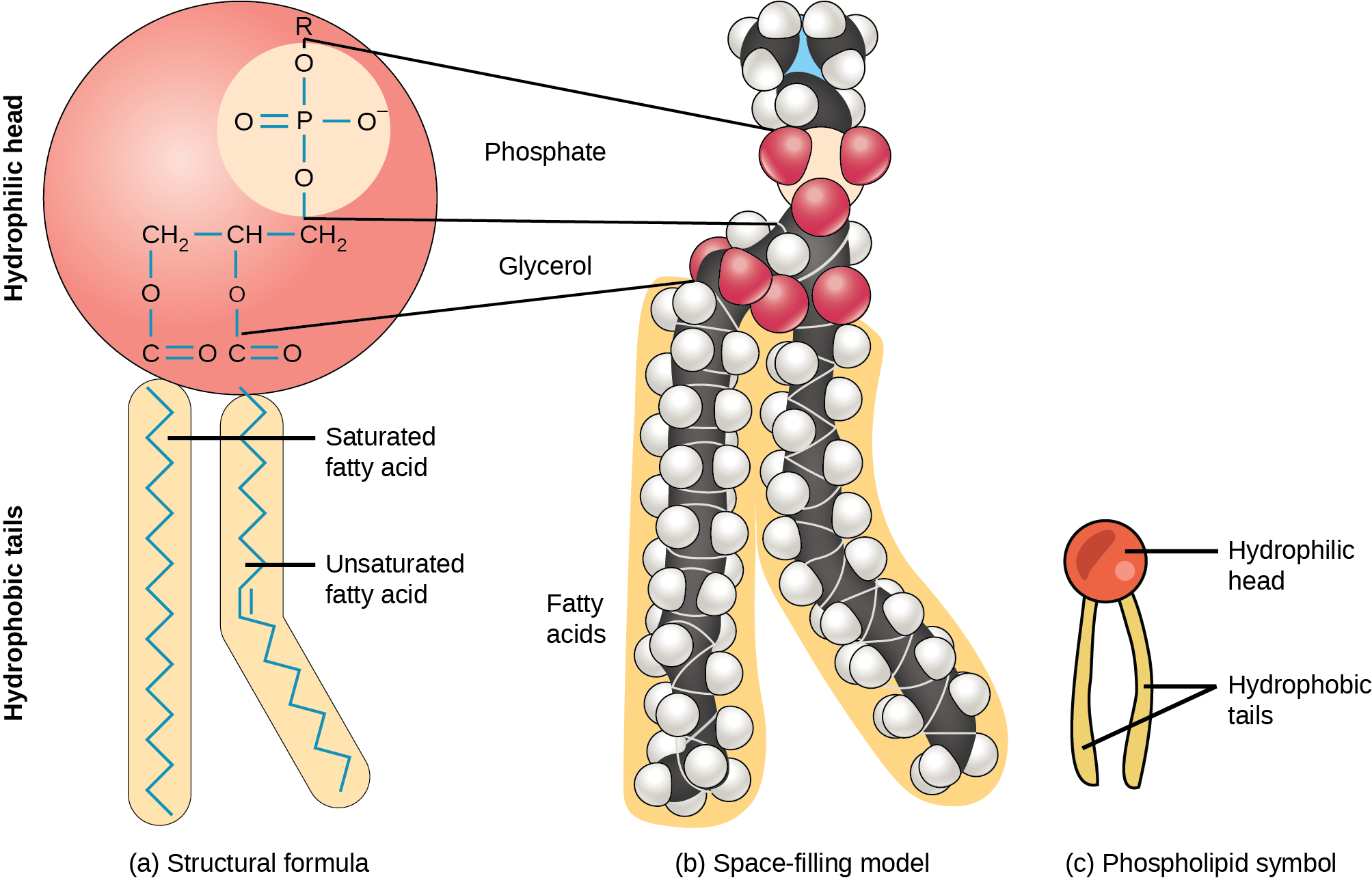
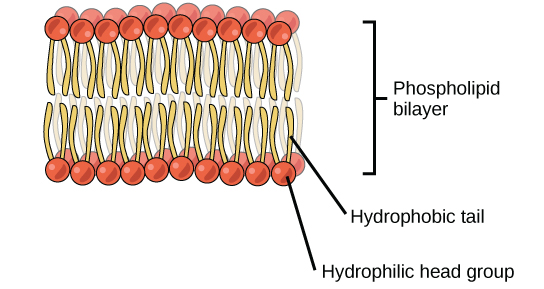
A phospholipid is an amphipathic molecule, meaning it has a hydrophobic and a hydrophilic part. The fatty acid chains are hydrophobic and cannot interact with water; whereas, the phosphate-containing group is hydrophilic and interacts with water. In aqueous solution, phospholipids can form a bilayer (Figure 4.17), where the fatty acid tails make up the hydrophobic core of the bilayer and the hydrophilic groups face the aqueous exterior. The phospholipid bilayer forms the basis of biological membranes.
A single layer of phospholipids form a spheroidal micelle rather than a bilayer. The phospholipids of the micelle are oriented with the tails inside the hydrophobic centre and the phosphate groups on the surface of the micelle in contact with the aqueous exterior.
Steroids
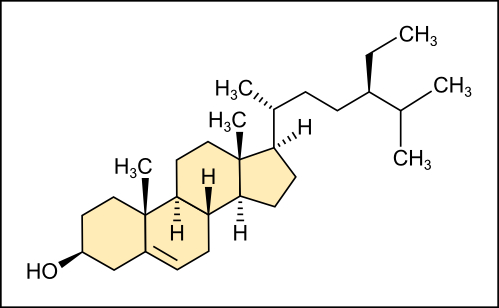
Unlike the fats and phospholipids that we discussed earlier, steroids have a fused ring structure. Although they do not resemble the other lipids, scientists consider steroids as lipids because steroids are also hydrophobic and insoluble in water. All steroids have four linked carbon rings and several of them, like cholesterol, have a short tail (Figure 4.18). Many steroids also have the –OH functional group, which puts them in the alcohol classification (sterols).
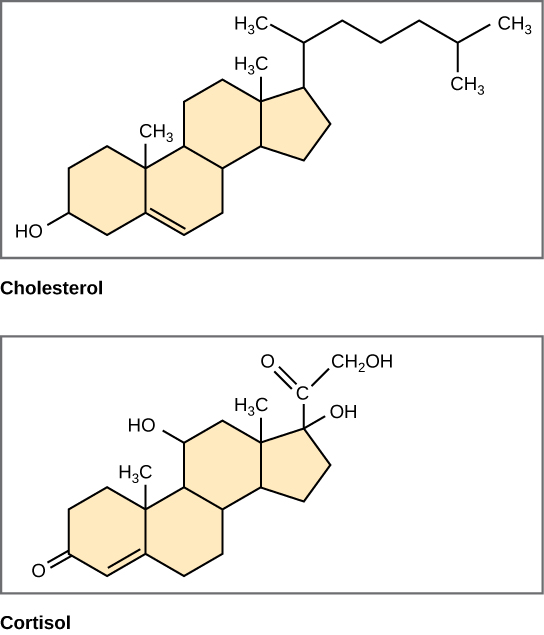
Cholesterol is the most common steroid. The liver synthesizes cholesterol and is the precursor to many steroid hormones such as testosterone and estradiol, which gonads and endocrine glands secrete. Cholesterol is also the precursor to vitamin D and bile salts, which help emulsifying fats and their subsequent absorption by cells. Although lay people often speak negatively about cholesterol, it is necessary for the body’s proper functioning. Sterols (cholesterol in animal cells, phytosterol in plants (Figure 4.19)) are components of the plasma membrane of cells and are found within the hydrophobic core of the phospholipid bilayer.
Section Summary
Lipids are a class of macromolecules that are nonpolar and hydrophobic in nature. Major types include fats and oils, waxes, phospholipids, and steroids. Fats are a stored form of energy and are also known as triacylglycerols or triglycerides. Fats are comprised of fatty acids and either glycerol or sphingosine. Fatty acids may be unsaturated or saturated, depending on the presence or absence of double bonds in the hydrocarbon chain. If only single bonds are present, they are saturated fatty acids. Unsaturated fatty acids may have one or more double bonds in the hydrocarbon chain. Phospholipids comprise the membrane’s matrix. They have a glycerol or sphingosine backbone to which two fatty acid chains and a phosphate-containing group are attached. Steroids are another class of lipids. Their basic structure has four fused carbon rings. Cholesterol is a type of steroid and is an important constituent of the plasma membrane, where it helps to maintain the membrane’s fluid nature. It is also the precursor of steroid hormones such as testosterone.
LINK TO LEARNING
For an additional perspective on lipids, explore the interactive animation “Biomolecules: The Lipids”.
Exercises
Glossary
lipid: macromolecule that is nonpolar and insoluble in water
omega fat: type of polyunsaturated fat that the body requires; numbering the carbon omega starts from the methyl end or the end that is farthest from the carboxylic end
phospholipid: membranes’ major constituent; comprised of two fatty acids and a phosphate-containing group attached to a glycerol backbone
saturated fatty acid: long-chain hydrocarbon with single covalent bonds in the carbon chain; the number of hydrogen atoms attached to the carbon skeleton is maximized
steroid: type of lipid comprised of four fused hydrocarbon rings forming a planar structure
trans fat: an isomer of unsaturated fatty acid where the carbon chain crosses to the opposite side of the carbon-carbon double bond
triacylglycerol (also, triglyceride): fat molecule; consists of three fatty acids linked to a glycerol molecule
unsaturated fatty acid: long-chain hydrocarbon that has one or more double bonds in the hydrocarbon chain
wax: lipid comprised of a long-chain fatty acid that is esterified to a long-chain alcohol; serves as a protective coating on some feathers, aquatic mammal fur, and leaves
Media Attributions
- Figure 4.9 by Ken Bosma
- Figure 4.15 by Roger Griffith
- Figure 4.19 by Jung-Lynn Jonathan Yang

